 we do not use proton excess or deficiency terminologies because protons are way more heavier than electrons and do not move when we charge a body either negative or positive. Anions are formed when an atom gains electrons. 'deficiency of electrons results in positive charge on body, while excess of electrons means body have negative charge. Anions always have less number of protons than electrons. The difference between a cation and an anion is the net electrical charge of the ion. hranions are negative ions and cations are positive ions.  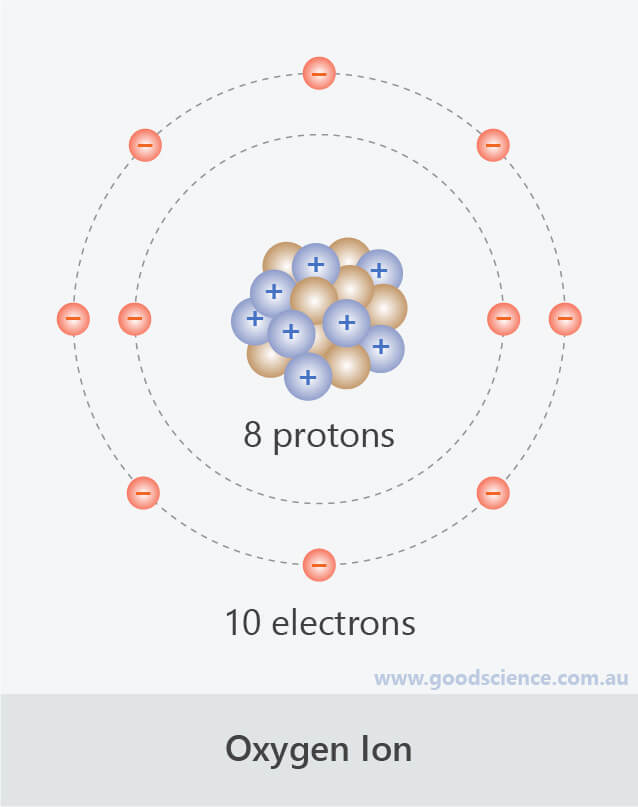
Trending Questions What is supernatured in Chemistry? How did Rene Descartes influence enlightenment astronomy? How does a Nebula affect us yes? What company did Sally Ride found? Which variable stars have pulsation periods between 1.5 hours and 1. Proton is the positively charged atom (+) while electron is the negatively charged atom (-). 100 (2 ratings) Transcribed image text: Question 12 Which of the following statements about ions is INCORRECT Cations always have more number of electrons than protons. Or, if an oxygen atom looses an electron then it becomes a positively charged ion because it has more positive protons than it does negative electrons. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
